
A new approach to Predator-prey dynamics
A note on predator-prey dynamics in radiocarbon datasets
Abstract
Recommendation: posted 29 February 2024, validated 02 March 2024
Blasco, R. (2024) A new approach to Predator-prey dynamics. Peer Community in Archaeology, 100458. https://doi.org/10.24072/pci.archaeo.100458
Recommendation
Various biological systems have been subjected to mathematical modelling to enhance our understanding of the intricate interactions among different species. Among these models, the predator-prey model holds a significant position. Its relevance stems not only from its application in biology, where it largely governs the coexistence of diverse species in open ecosystems, but also from its utility in other domains.
Predator-prey dynamics have long been a focal point in population ecology, yet access to real-world data is confined to relatively brief periods, typically less than a century. Studying predator-prey dynamics over extended periods presents challenges due to the limited availability of population data spanning more than a century. The most extensive dataset is the hare-lynx records from the Hudson Bay Company, documenting a century of fur trade [1]. However, other records are considerably shorter, usually spanning decades [2,3]. This constraint hampers our capacity to investigate predator-prey interactions over centennial or millennial scales.
Marom and Wolkowski [4] propose here that leveraging regional radiocarbon databases offers a solution to this challenge, enabling the reconstruction of predator-prey population dynamics over extensive timeframes. To substantiate this proposition, they draw upon examples from Pleistocene Beringia and the Holocene Judean Desert. This approach is highly relevant and might provide insight into ecological processes occurring at a time scale beyond the limits of current ecological datasets.
The methodological approach employed in this article proposes that the summed probability distribution (SPD) of predator radiocarbon dates, which reflects changes in population size, will demonstrate either more or less variation than anticipated from random sampling in a homogeneous distribution spanning the same timeframe. A deviation from randomness would imply a covariation between predator and prey populations. This basic hypothesis makes no assumptions about the frequency, mechanism, or cause of predator-prey interactions, as it is assumed that such aspects cannot be adequately tested with the available data. If validated, this hypothesis would offer initial support for the idea that long-term regional radiocarbon data contain signals of predator-prey interactions. This approach could justify the construction of larger datasets to facilitate a more comprehensive exploration of these signal structures.
References
[1] Elton, C. and Nicholson, M., 1942. The Ten-Year Cycle in Numbers of the Lynx in Canada. J. Anim. Ecol. 11, 215–244.
[2] Gilg, O., Sittler, B. and Hanski, I., 2009. Climate change and cyclic predator-prey population dynamics in the high Arctic. Glob. Chang. Biol. 15, 2634–2652. https://doi.org/10.1111/j.1365-2486.2009.01927.x
[3] Vucetich, J.A., Hebblewhite, M., Smith, D.W. and Peterson, R.O., 2011. Predicting prey population dynamics from kill rate, predation rate and predator-prey ratios in three wolf-ungulate systems. J. Anim. Ecol. 80, 1236–1245. https://doi.org/10.1111/j.1365-2656.2011.01855.x
[4] Marom, N. and Wolkowski, U. (2024). A note on predator-prey dynamics in radiocarbon datasets, BioRxiv, 566733, ver. 4 peer-reviewed and recommended by Peer Community in Archaeology. https://doi.org/10.1101/2023.11.12.566733
The recommender in charge of the evaluation of the article and the reviewers declared that they have no conflict of interest (as defined in the code of conduct of PCI) with the authors or with the content of the article. The authors declared that they comply with the PCI rule of having no financial conflicts of interest in relation to the content of the article.
European Research Council DEADSEA_ECO ERC-Stg grant (#802752)
Evaluation round #2
DOI or URL of the preprint: https://doi.org/10.1101/2023.11.12.566733
Version of the preprint: 2
Author's Reply, 18 Feb 2024
Dear Dr. Blasco,
Thank you and the reviewers for your additional comments.
1. Regarding Dr. Rodrigues' point:
We appreciate the observation that the simulated predator dates are drawn from a uniform (homogeneous) distribution. Consequently, the statistic we display quantifies the distance from the prey SPD to SPDs drawn from a random distribution. We fully agree with this assessment.
Our approach avoids making assumptions about the specific nature of the predator-prey periodicity, as we lack prior knowledge. Hence, we neither assume that predator cycles should closely match prey cycles (by positively weighting the probability of predator random sampling based on the prey SPD) nor that they exhibit a complete phase shift (by negatively weighting the probability). Instead, we adopt the most parsimonious and minimalist hypothesis, suggesting that some non-random (non-uniform) relationship likely exists.
Following the reviewer's comment, we have clarified the point regarding the homogeneous distribution on page 3, paragraph 3. The revised text now reads:
"We hypothesize that the summed probability distribution (SPD) of predator radiocarbon dates, insofar as it tracks changes in population size, will have either greater or lesser divergence than expected from random SPDs sampling in a homogeneous distribution from the same time range."
2. Species names in the methods section:
We have italicized the species names in the methods section, as you suggested.
Respectfully,
Nimrod & Uri
Decision by Ruth Blasco , posted 17 Feb 2024, validated 18 Feb 2024
, posted 17 Feb 2024, validated 18 Feb 2024
After careful consideration of your revised manuscript, I'm pleased to inform you that the reviewers agree that the manuscript has improved substantially. They find the innovative approach to investigating predator-prey dynamics in deep time to be a relevant topic deserving of publication. However, before final acceptance, there is a minor revision suggested by Reviewer 1 that requires attention.
Reviewer 1 noted an interesting point regarding the methodology section. It would be beneficial to address the clarification regarding the selection probability of integer values within the range of radiocarbon ages of the prey. Please carefully review his comment and ensure clarity on this aspect in your revision.
Additionally, please remember to italicize "Hyaena hyaena" and "Gazella dorcas" in the first paragraph of the Methods section.
Thank you for your diligence and effort in addressing the reviewers' comments and making the necessary revisions for publication. With the inclusion of these adjustments, I am confident that your manuscript will be accepted in the next round of review.
Reviewed by Jesús Rodríguez, 07 Feb 2024
In the revised version of the manuscript and in their reply, the authors adressed all my previous comments and criticism and provide reasoned answers to them. I must confess, however, that I am not entirely convinced by all their arguments, but this should not prevent publication of the manuscript. The authors present an innovative approach to investigate predator-prey dynamics in deep time, and this is a relevant topic. Thus, I consider that the manuscript should be published so its controversial points may be discussed by the scientific community.
I would like, however, to point out a last comment. The basis of the methodology is to compare the observed divergence between the SPD of the prey and the predator with the divergence between the SPD of the prey and “… a random set of integers, equivalent to the sample size of the predator, (which is)… drawn from the range of the radiocarbon years of the prey”. According to this, all the integer values inside the range of radiocarbon ages of the prey have the same probability of being selected. This is equivalent to say that the random sample is drawn from a homogenous distribution of radiocarbon dates from the range of radiocarbon years of the prey. If I am correct, what the method is testing is whether the observed divergence between the SPD of the prey and the SPD of the predator is different from the divergence between the observed SPD of the prey and an homogeneous distribution.
https://doi.org/10.24072/pci.archaeo.100458.rev21Reviewed by anonymous reviewer 1, 15 Feb 2024
Dear Dr. Blasco,
I believe the authors have made changes to the text that have strengthened it, and I do not have much to add except that in the first paragraph of the Methods section, Hyaena hyaena and Gazella dorcas appear without italics.
Sincerely yours
Evaluation round #1
DOI or URL of the preprint: https://doi.org/10.1101/2023.11.12.566733
Version of the preprint: 1
Author's Reply, 01 Feb 2024
Decision by Ruth Blasco , posted 23 Jan 2024, validated 23 Jan 2024
, posted 23 Jan 2024, validated 23 Jan 2024
We have received comments from three reviewers on your manuscript. While they find your work interesting, they have raised important points that need to be addressed in a revision. Some of the main concerns involve a more in-depth explanation of assumptions and hypotheses, as well as greater detail and justification of the methods used. A crucial point is the need to address the taphonomic problem that entails a higher or lesser representation of species in the sites. Please keep in mind that, in addition to the behavior of a given species, the taphonomic characteristics of the site also affect the probability of finding its remains in the fossil record. This issue needs consideration in the article and further development, as it can directly impact the results and their interpretation. Another important point is to provide a detailed justification for the assumption that the SPD of radiocarbon dates of predators is different from those of prey.
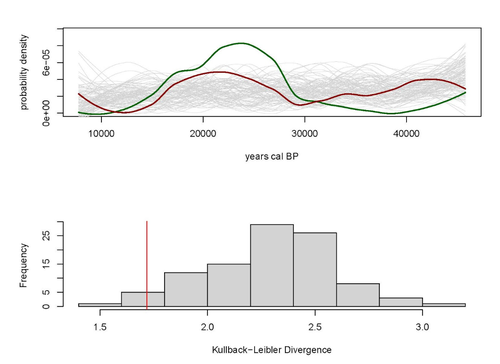
Regarding the two test cases selected by the authors to illustrate their approach (Fairbanks and Judean Desert datasets), it seems that they do not fully meet some of the requirements proposed initially by the authors. The authors argue that the radiocarbon datasets required should be spatially constrained and include randomly collected specimens. Please elaborate more on how the selection was made and justify in detail their inclusion in the study. Finally, it would also be useful to explain how to interpret the results of Kullback-Leibler divergence analysis, including figures 1 and 2, and when two distributions can be considered not different.
I hope that the reviewers' comments prove helpful and contribute to strengthening your manuscript, as your approach holds the potential to provide valuable information about ecological processes occurring on a timescale beyond the limits of current ecological datasets.
Reviewed by Jesús Rodríguez, 21 Dec 2023
The preprint “A note on predator-prey dynamics in radiocarbon datasets” by N. Maron and U. Wolkowski addresses a very relevant topic with a novel approach. The authors focus on predator-prey dynamics on a millennial time scale, using datasets composed of radiocarbon dates of single fossils. This is much longer than the period usually covered by the datasets used in the ecological literature, and might potentially shed light on ecological processes occurring over extended periods. Although I am sympathetic to the aim of the authors, I found the explanation of their assumptions and hypotheses insufficient and their methods difficult to follow. Moreover, I detected some flaws in the datasets used to illustrate their approach, which might compromise the validity of their conclusions. I elaborate on these topics below and suggest ways to improve their approach.
The basis of the authors’ approach is an adaptation to Palaeontology of the so-called “dates as data” paradigm of Archaeology, “…which assumes that the number of radiocarbon dates in a region reflects the magnitude of occupation or the total number of person-years of human existence.” This paradigm is transferred to Palaeontology by the authors by stating that “… the probability of a specimen surviving to be dated is assumed to be proportional to the number of individuals of its taxon that existed in a specific region and time.” This is, in my opinion, the main flaw of their approach. I agree that the population density of a species in a limited area is one of the main factors affecting the probability of finding their remains in the paleontological record and obtaining radiocarbon dates for them. However, other interacting factors can drastically modify the probability of finding a certain taxon in a fossil assemblage. Biotic and abiotic accumulation agents introduce biases that modify the probability of finding the remains of a certain taxon, independently of its population density. Just as an example, predators are selective and select their prey mainly by body size. Thus, two predators largely differing in size hunting in the same area produce assemblages with different taxonomic compositions. The behaviour of a species, in combination with the characteristics of the site, also affects the probability of finding its remains in the fossil record. Carnivores denning in caves are usually overrepresented in cave sites with respect to their density in the environment. In contrast, the presence of ungulates in those sites is usually due to the activity of the carnivores, or humans, and their abundance is biased by the predator’s preferences. Post-depositional processes may also affect differentially the skeletal elements of different taxa, making a species abundant in the environment to be rarer in a fossil assemblage than other species whose population density was lower.
The hypothesis tested by the authors is that the summed probability distribution (SPD) of radiocarbon dates of predators is different from those of prey. The authors state that if differences exist, they prove the existence of a signal of predator-prey interactions in the analysed datasets. I found this assumption insufficiently justified in the preprint. It may be correct, but the connection between the differences in the SPDs of predators and prey and the signal of interactions among them over time is not evident to me. I would ask the authors to elaborate on this topic, which may be highly relevant, and do not assume that the connection is evident to everyone.
I have relevant concerns about the suitability of the two test cases selected by the authors to illustrate their approach, namely the Fairbanks and Judean Desert Datasets. It is stated by the authors that the radiocarbon datasets required by their approach should be spatially constrained and include randomly collected specimens. Unfortunately, there are reasons to think that the two selected datasets do not meet the later criterion.
The Fairbanks dataset was compiled by Leonard et al. (2007) and reused by Fox-Dobbs et al. (2008) and includes radiocarbon dates of fossils found in mining gravel deposits in the Fairbanks area. The Fairbanks dataset contains radiocarbon dates for 40 fossils of wolves, 32 fossils of horses and 10 fossils of reindeer. However, only 33 fossils of wolves, 28 fossils of horses and 6 fossils of reindeer are included in the preprint. I guess that the authors excluded the 7 wolves, 4 horses and 4 reindeer with only minimum age estimates (Leonard et al., 2007 Supplemental Data Table S1). The decision is correct, but maybe the authors should mention it in the text. My major concern is, however, about the random selection of the fossils included in the dataset and their representativity of the past living communities. The dataset is biased towards an overrepresentation of the predator. Predators occur always at much lower population densities than their prey (Damuth, 2007) because in any ecosystem the biomass of a predator is 100 times lower than the biomass of its prey (Carbone and Gittleman, 2002). In contrast, the Fairbanks dataset contains more wolves than horses and 5 times more wolves than reindeer. Thus, the dataset violates the assumption that the probability of a specimen surviving to be dated is proportional to the population density of the species.
The Judean Desert dataset provides radiocarbon dates for fossils of seven species of large mammals collected in 43 caves (Lazagabaster et al., 2022). It includes 14 dated specimens of Sus scrofa, 10 of Capra nubiana, 15 of Gazella spp, 25 of Procavia capensis, 20 of Hyaena hyaena, 12 of Panthera pardus, and 14 of Vulpes spp. Maron and Wolkowski selected three taxa from this list for their test analysis (Capra nubiana, Procavia capensis, and Panthera pardus) and do not mention the existence of data about the other taxa excluded from their analysis. I guess that they selected the data for a predator (P. pardus) and its potential prey (C. nubiana and P. capensis), since their goal is the relationship between predators and their prey. However, this choice should be reported and explained. Why did the authors exclude Sus scrofa and Gazella spp from the analysis? If it is because they are not considered potential prey of the leopard, this should be stated and justified. Moreover, carnivores are overrepresented in the Judean dataset as they are in the Fairbanks dataset. All taxa are represented in the Judean dataset by a similar number of specimens (between 10 and 25), though the population densities of hyenas and leopards in the past ecosystems may be expected to be one or two orders of magnitude lower than those of the other taxa, as explained above. Furthermore, the Judean Desert dataset illustrates other of the flaws of this approach. Maron and Wolkowski included in their analysis the radiocarbon dates for the 12 specimens of P. pardus reported by Lazagabaster et al. (2022). However, these 12 specimens were found in 3 caves and they correspond to a minimum number of only six individuals (Lazagabaster et al., 2022). Thus, there is an obvious problem of pseudoreplication in the data. Moreover, as stated by the authors, to comply with the requirements of the “dates as data” paradigm the datasets should include randomly selected specimens. This means that the specimens to be dated should be selected at random from the available fossil assemblages, but the Judean desert dataset violates this requirement. Lazagabaster et al. (2022) wrote on page 654: “We prioritized dating and isotopic analysis of the large (>21 kg) carnivores P. pardus nimr and H. hyaena, acknowledging their role as keystone taxa and their relative rarity in the mammal community.” Thus, Lazagabaster et al. (2022) consciously overrepresented the specimens of carnivores in their dataset of radiocarbon dates. This illustrates other of the potential flaws of the approach. Since the radiocarbon dates are not obtained with the aims of the “dates as data” paradigm in mind, the authors of the original source may introduce biases necessary for their own goal in selecting specimens to be dated.
The methods are not properly explained. It is stated in the introduction that the hypothesis to be tested is that the SPD of predator radiocarbon dates is different from the SPDs of the prey. To test this hypothesis the authors use the Kullback-Leiber (KL) measure of divergence and estimate the probability of obtaining by chance, through a Monte Carlo approach, a value of divergence as the one observed in the dataset. In the results section, the authors report that the KL divergence for the Fairbanks dataset is “smaller than 98% of the divergences measured for (random) predator-(real) prey distributions”. To my knowledge, this means that the divergence is too small to be produced by chance, and thus, that the SPDs of the predator and the prey do not diverge. Surprisingly, the authors conclude that “This supports our hypothesis that the Fairbanks predator and prey distributions are not random and that the low divergence… is therefore unlikely to be due to chance”. However, the hypothesis stated in the introduction was that the SPDs are different.
The interpretation made by the authors of the results for the Judean Desert Dataset is also disconcerting for me. In this case, the KL divergence value is said to be “smaller than 94% of the divergences measured for (random) predator – (real) prey distributions”. Again, the authors conclude that the “…predator and prey distributions are not random, and that the low divergence between them is, therefore, unlikely to be due to chance.” However, figure 2 shows that the observed KL value is not smaller but larger than most of the values generated at random. Thus, if the KL value observed is within 95% of the values obtained in the Monte Carlo approach, the conclusion should be that the KL value observed may be obtained by chance. Only if the value observed in the dataset is larger than 95% of the randomly produced values it is possible to conclude that the divergence observed is too large to be a result of chance. In any case, a theoretical justification of the meaning of a similarity or divergence between the SPDs of predators and prey is required.
In summary, I would recommend the authors revise their analyses and produce a reviewed and expanded version of the manuscript. They should provide a theoretical background to their main hypothesis, explaining the connection between a signal of the predator-prey dynamics at the millennial scale and the SPDs of the radiocarbon dates of predators and prey. Moreover, I recommend the authors critically review the effects of different factors on the signal and how the accumulation and post-depositional processes may affect it. It would be very useful also to discuss possible solutions to avoid biases in the data. For example, select fossils from assemblages accumulated by similar agents and with comparable post-depositional processes. The methods should be better explained as well as the interpretation of the results.
The question addressed in this preprint is highly relevant. If successful, this approach might provide insight into important ecological processes occurring at a time scale beyond the limits of current ecological datasets. Thus, it deserves attention and careful consideration.
Minor comments
-The authors wrote that the Fairbanks database records “regional mortality between ~40-12 kya” (line 75). However, it includes a specimen dated to 7751 + 64 kya (Supplementary Table 1).
-There is a typo in line 78. Capra ibex nubiana, not “Cara ibex nubiana”
-Line 68. The acronym SPD is not defined until line 97, it should be spelled here.
References
Carbone, C., Gittleman, J.L., 2002. A common Rule for the Scaling of Carnivore Density. Science 295, 2273-2275.
Damuth, J., 2007. A Macroevolutionary Explanation for Energy Equivalence in the Scaling of Body Size and Population Density. The American Naturalist 169, 621-631.
Fox-Dobbs, K., Leonard, J., Koch, P., 2008. Pleistocene megafauna from eastern Beringia: Paleoecological and paleoenvironmental interpretations of stable carbon and nitrogen isotope and radiocarbon records. Palaeogeography, Palaeoclimatology, Palaeoecology 261, 30-46. 10.1016/j.palaeo.2007.12.011
Lazagabaster, I.A., Égüez, N., Ullman, M., Porat, R., Wachtel, I., Davidovich, U., Marom, N., 2022. Cave paleozoology in the Judean Desert: assembling records of Holocene wild mammal communities. Journal of Quaternary Science 37, 651-663. https://doi.org/10.1002/jqs.3405
Leonard, J.A., Vilà, C., Fox-Dobbs, K., Koch, P.L., Wayne, R.K., Van Valkenburgh, B., 2007. Megafaunal Extinctions and the Disappearance of a Specialized Wolf Ecomorph. Current Biology 17, 1146-1150. https://doi.org/10.1016/j.cub.2007.05.072
https://doi.org/10.24072/pci.archaeo.100458.rev11
Reviewed by anonymous reviewer 1, 10 Jan 2024
Reviewed by Miriam Belmaker, 16 Jan 2024
This is an elegant paper addressing a problem related to neoecological research and archaeology. The question of predator-prey interactions has been a staple in ecological research. As this paper shows, the non-random distribution of predator and prey is an excellent starting point for discussing the red queen and court jester forces in the temporal and spatial distribution of fauna over time. Ideally, future research would also be able to incorporate climatic data. New advances in paleothermometers may allow these correlations in the future.
One comment - using red and green lines in the figures is not conducive to color-blind readers, and I recommend finding alternative colors.
https://doi.org/10.24072/pci.archaeo.100458.rev13









